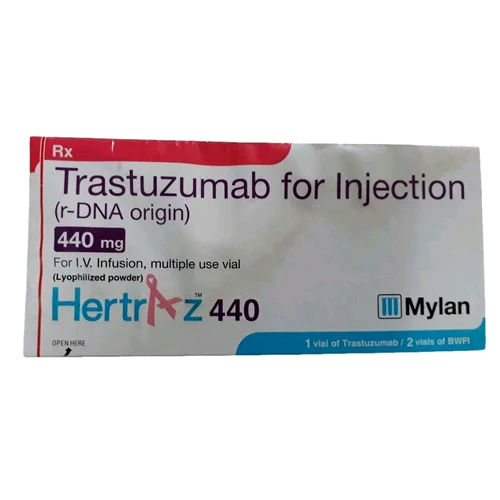
Hertraz 440 mg Injection
Hertraz 440 mg Injection Specification
- Dosage Form
- Lyophilized Powder for Injection
- Indication
- Breast cancer, Metastatic gastric cancer
- Salt Composition
- Trastuzumab (440mg)
- Enzyme Types
- Not an enzyme (Monoclonal antibody)
- Feature
- Recombinant humanized monoclonal antibody targeting HER2 receptor
- Ingredients
- Trastuzumab, excipients and sterile water for injection (as solvent)
- Application
- Other
- Ph Level
- As per company norms
- Physical Color/Texture
- White to pale yellow lyophilized powder
- Fermentation Smell
- Normal Smell
- Storage Instructions
- Dry place
- Shelf Life
- upto 24 Months
- Contraindications
- Hypersensitivity to trastuzumab or any excipients
- Method of Preparation
- Reconstitute with provided solvent before use
- Usage Precaution
- Monitor cardiac function during therapy
- Suitable For
- Adult patients
- Pack Size
- 1 vial + 1 solvent ampoule
- Target Receptor
- HER2/neu receptor
- Dispensing Condition
- To be administered by healthcare professional only
- Regulatory Approval
- DCGI approved
- Administration Route
- Intravenous infusion
- Prescription Status
- Prescription only
- Solvent Provided
- Yes, sterile water for injection included
- Packaging Type
- Box pack with vial and ampoule
- Strength
- 440 mg per vial
Hertraz 440 mg Injection Trade Information
- Minimum Order Quantity
- 1 Unit
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 5000 Units Per Month
- Delivery Time
- 7-10 Seconds
- Main Export Market(s)
- Asia, Australia, North America, Eastern Europe, Western Europe
- Main Domestic Market
- All India
About Hertraz 440 mg Injection
Hertraz 440 mg Injection: Areas of Application & Key Features
Hertraz 440 mg Injection is specially indicated for adjuvant and metastatic breast cancer, as well as metastatic gastric cancer. Its major application areas lie in targeting HER2/neu receptors, providing patients with advanced therapy options. Key features include recombinant monoclonal antibody formulation, easy reconstitution with supplied sterile water, and suitability for adult patients. The injection comes as a white to pale yellow lyophilized powder, ensuring precise dosage. Each pack includes a vial and solvent ampoule, offering exceptional reliability.
Hertraz 440 mg: Market Availability & Packaging Insights
Hertraz 440 mg Injection is in stock ready for immediate dispatch, meeting significant demands across the main domestic market in India. Each unit is packaged securely-one vial and one solvent ampoule per box. Sample policy is flexible; you may request a sample before deciding the final asking price or estimate for bulk orders. The packaging ensures safety and maintains the product's shelf life and quality, giving confidence to both wholesalers and exporters.
Specification
|
Directions for Use |
For I.V Infusion,Multiple Use Vial |
|
Form |
Injection |
|
Packaging Size |
1 Vial of Trastuzumab,2 Vial Of BWFI |
|
Prescription/Non prescription |
Prescription |
|
Packaging |
Vial |
|
Treatment |
Adjuvant breast cancer,Metastatic breast cancer,Metastatic gastric cancer. |
|
Manufacturer |
Mylan |
|
Composition |
Trastuzumab |
|
Brand |
Hertraz 440 |
FAQ's of Hertraz 440 mg Injection:
Q: How should Hertraz 440 mg Injection be prepared and administered?
A: Hertraz 440 mg Injection must be reconstituted with the supplied sterile water for injection before intravenous infusion. Administration should be done by a qualified healthcare professional, following all recommended guidelines for dosage and infusion rate.Q: What is the main indication for using Hertraz 440 mg Injection?
A: The injection is primarily indicated for adult patients with breast cancer (adjuvant and metastatic) and metastatic gastric cancer where HER2/neu receptor expression is confirmed.Q: When should cardiac function be monitored during Hertraz therapy?
A: Cardiac function should be closely monitored before starting, during, and after Hertraz therapy, as there can be cardiac-related risks associated with Trastuzumab treatment.Q: Where should Hertraz 440 mg Injection be stored?
A: Store Hertraz 440 mg Injection in a dry place, away from direct sunlight, adhering to company norms and keeping it under recommended temperature conditions to preserve efficacy.Q: What are the benefits of choosing Hertraz 440 over alternatives?
A: Hertraz 440 mg Injection provides a DCGI-approved, quality-assured treatment with a prodigious safety and reliability profile, and a value-based price point, making it a trusted solution in cancer therapy.Q: What precautions should be observed before starting treatment with Hertraz?
A: Before initiating therapy with Hertraz, ensure the patient does not have hypersensitivity to Trastuzumab or its excipients, and assess baseline cardiac function due to possible adverse cardiac effects.
Price 57500 INR/ Unit
- Minimum Order Quantity
- 1 Unit
- Supply Ability
- 5000 Units Per Month
- Delivery Time
- 7-10 Seconds
- Main Domestic Market
- All India
- Main Export Market(s)
- Asia, Australia, North America, Eastern Europe, Western Europe

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Anti Cancer Injection Category
440 MG Trastuzumab For Injection
Price 14000 INR / Unit
Minimum Order Quantity : 1 Unit
Physical Color/Texture : Tan liquid
Fermentation Smell : Normal Smell
Shelf Life : 36 months (unopened vial)
Storage Instructions : Dry place
Borviz 2.5 Mg Injection
Price 1000 INR / Unit
Minimum Order Quantity : 1 Unit
Physical Color/Texture : Tan liquid
Fermentation Smell : Normal Smell
Shelf Life : upto 24 Days
Storage Instructions : Dry place
Nandrix - Phenlypropionate 100 mg-ml
Price 200 INR / Vial
Minimum Order Quantity : 10 Vials
Physical Color/Texture : Tan liquid
Fermentation Smell : Normal Smell
Shelf Life : upto 24 Months
Storage Instructions : Dry place
Oncotrex 50mg Injection
Price 1000 INR / Unit
Minimum Order Quantity : 10 Units
Physical Color/Texture : Tan liquid
Fermentation Smell : Normal Smell
Shelf Life : upto 24 Days
Storage Instructions : Dry place