Biceltis Trastuzumab Injection
Biceltis Trastuzumab Injection Specification
- Usage
- Hospital
- Origin of Medicine
- India
- Pacakaging (Quantity Per Box)
- 1 vial per box
- Indication
- HER2-overexpressing breast and gastric cancers
- Expiration Date
- 24-36 months from manufacturing date
- Molecular Formula
- C6470H10012N1726O2013S42
- Salt Composition
- Trastuzumab
- Medicine Type
- Injection
- Dosage
- 440 mg/vial
- Dosage Form
- Lyophilized Powder for Reconstitution
- Storage
- Dry Place
- Appearance
- Liquid
- Grade
- Pharma
- CAS No
- 180288-69-1
- Assay
- 98%
- Route of Administration
- Intravenous infusion
- Reconstitution Requirement
- Requires reconstitution with sterile Water for Injection
- Preservative
- No preservative
- Adverse Reactions
- Infusion reactions, cardiotoxicity, pulmonary toxicity
- Container Type
- Single-use glass vial
- Product Registration No.
- As per Indian drug regulations
- Contraindication
- Hypersensitivity to Trastuzumab
- Brand Name
- Biceltis
- Prescription Status
- Prescription Only (Rx)
- Therapeutic Class
- Monoclonal Antibody
- Compatibility
- Do not mix with other drugs
- Stability after Reconstitution
- Stable for 24 hours at 2C to 8C
Biceltis Trastuzumab Injection Trade Information
- Minimum Order Quantity
- 100 Packs
- Supply Ability
- 5000 Packs Per Month
- Delivery Time
- 7 Days
- Main Export Market(s)
- Asia, Australia, North America, Eastern Europe, Western Europe
About Biceltis Trastuzumab Injection
Biceltis Trastuzumab Injection contains the active ingredient trastuzumab, which is a monoclonal antibody used in the treatment of HER2-positive breast cancer and certain other HER2-positive cancers. It works by targeting and binding to the HER2 protein, which is overexpressed in HER2-positive cancers. This is typically administered intravenously (into a vein) under the supervision of a healthcare professional. By binding to HER2, trastuzumab inhibits the growth and proliferation of cancer cells and can also stimulate the immune system to attack cancer cells. Furthermore, you can avail the offer of Biceltis Trastuzumab Injection at very reasonable prices.
Specification
|
Form |
Injection |
|
Prescription/Non prescription |
Prescription |
|
Treatment |
To treat breast cancer |
|
Manufacturer |
Emcure |
|
Composition |
Trastuzumab |
|
Brand |
Biceltis |
Precision Therapy for Cancer Treatment
Biceltis Trastuzumab Injection stands at the forefront of precision medicine for HER2-overexpressing breast and gastric cancers. By targeting specific cancer cell receptors, Biceltis helps slow tumor growth, offering renewed hope to patients. Used under hospital supervision, it delivers focused treatment for better clinical outcomes.
Quality, Safety, and Stability You Can Trust
Each vial of Biceltis is manufactured in India adhering to strict pharmaceutical standards. Supplied as a single-use glass vial containing pure lyophilized powder, Biceltis maintains stability for up to 24 months and up to 24 hours after reconstitution when properly refrigerated. It guarantees both safety and efficacy for patient care.
FAQ's of Biceltis Trastuzumab Injection:
Q: How is Biceltis Trastuzumab Injection prepared and administered?
A: The lyophilized powder in the single-use vial must be reconstituted with sterile Water for Injection before use. After reconstitution, the solution is administered via intravenous infusion in a hospital setting by qualified healthcare professionals.Q: What are the main indications for Biceltis Trastuzumab Injection?
A: Biceltis is indicated for the treatment of HER2-overexpressing breast and gastric cancers, providing targeted therapy as part of a prescribed oncology regimen.Q: When should Biceltis not be used?
A: Biceltis is contraindicated in patients who have a known hypersensitivity to Trastuzumab or any of its components. Always inform your healthcare provider of any allergies before starting treatment.Q: Where should Biceltis be stored after reconstitution?
A: Once reconstituted, Biceltis should be stored between 2C and 8C and remains stable for 24 hours. The unused portion must be discarded after this period.Q: What is the process for reconstituting Biceltis Trastuzumab Injection?
A: To reconstitute, add sterile Water for Injection to the vial as per the instructions provided in the product leaflet. Gently swirl-do not shake-until fully dissolved. The resulting liquid is clear to slightly opalescent and ready for infusion.Q: What are some possible adverse reactions associated with Biceltis?
A: Patients may experience infusion reactions, cardiotoxicity, or pulmonary toxicity. Immediate medical supervision is advised during administration and for observation after infusion.Q: Why is it important not to mix Biceltis with other drugs during administration?
A: Biceltis should not be mixed with other medications to avoid incompatibility that may compromise its stability, efficacy, or safety profile.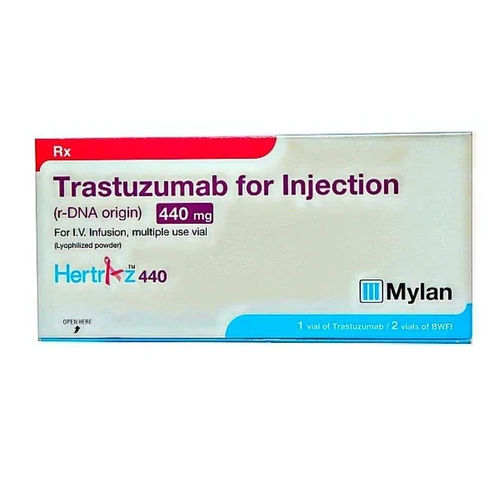
Price 75000 INR/ Box
- Minimum Order Quantity
- 100 Packs
- Supply Ability
- 5000 Packs Per Month
- Delivery Time
- 7 Days
- Main Export Market(s)
- Asia, Australia, North America, Eastern Europe, Western Europe

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Anti Cancer Drug Category
Velpanat 400 Mg100mg Tablets
Price 18000 INR / Box
Minimum Order Quantity : 100 Bottles
Appearance : Tablets
Dosage : 100 mg & 400 mg
Storage : Dry Place
Usage : Hospital
Erlocip 150 Mg Erlotinib Tablet
Price 10000 INR / Box
Minimum Order Quantity : 100 Bottles
Appearance : Tablets
Dosage : 150 mg
Storage : Dry Place
Usage : Hospital
Geffon-Gefitinib 250mgTablets
Price 5000 INR / Box
Minimum Order Quantity : 100 Boxes
Appearance : Tablets
Dosage : 250mg
Storage : Dry Place
Usage : Hospital
Cabozantinib 60 Mg Tablet
Price 7000 INR / Box
Minimum Order Quantity : 100 Bottles
Appearance : Tablets
Dosage : 60 mg
Storage : Dry Place
Usage : Hospital