440 MG Ttrastuzumab For Injection
440 MG Ttrastuzumab For Injection Specification
- Application
- Other
- Ph Level
- As per company norms
- Physical Color/Texture
- Tan liquid
- Fermentation Smell
- Normal Smell
- Storage Instructions
- Dry place
- Shelf Life
- upto 24 Months
440 MG Ttrastuzumab For Injection Trade Information
- Minimum Order Quantity
- 100 Units
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 5000 Units Per Month
- Delivery Time
- 7-10 Seconds
- Main Export Market(s)
- Asia, Australia, North America, Eastern Europe, Western Europe
- Main Domestic Market
- All India
About 440 MG Ttrastuzumab For Injection
Premium Quality and Stability
Trastuzumab for Injection, available in 440 MG strength, maintains its stability for up to 24 months when stored correctly. Its tan-colored liquid form, combined with normal fermentation aroma and strict compliance to pH standards, demonstrates its quality and careful production process to meet clinical needs.
Safe Storage and Handling
To preserve the potency and effectiveness of this trastuzumab formulation, it must be stored in a dry place, away from moisture and direct sunlight. Following these storage instructions extends shelf life and reduces the risk of contamination, ensuring that each dose retains its full therapeutic potential.
FAQ's of 440 MG Ttrastuzumab For Injection:
Q: How should 440 MG Trastuzumab for Injection be stored to ensure maximum shelf life?
A: This medication should be stored in a dry place to reach its maximum shelf life of up to 24 months. Avoid exposing the product to moisture, heat, or direct sunlight.Q: What is the physical appearance and smell of 440 MG Trastuzumab for Injection?
A: The injection appears as a tan liquid and possesses a normal fermentation smell, indicating its standard and unaltered composition.Q: When should 440 MG Trastuzumab for Injection be administered?
A: Trastuzumab should be administered according to a healthcare provider's instructions, typically in a hospital or clinical setting. The timing and dosage depend on the patient's specific treatment protocol.Q: Where can I source 440 MG Trastuzumab for Injection?
A: This medication is available for export and wholesale through suppliers based in India, ensuring broad accessibility for medical professionals and organizations.Q: How is the pH level of the product maintained?
A: The pH of 440 MG Trastuzumab for Injection is meticulously maintained as per company norms, ensuring the product's integrity, safety, and effectiveness during storage and administration.Q: What is the benefit of this trastuzumab formulation for patients?
A: This trastuzumab injection is used for targeted therapy in HER2-positive cancers, offering significant clinical benefits such as inhibiting tumor growth and improving treatment outcomes when used as directed by medical professionals.Q: What is the process for using this injectable medication?
A: A qualified healthcare professional will reconstitute and administer this medication through infusion, following established medical protocols to ensure patient safety and therapeutic efficacy.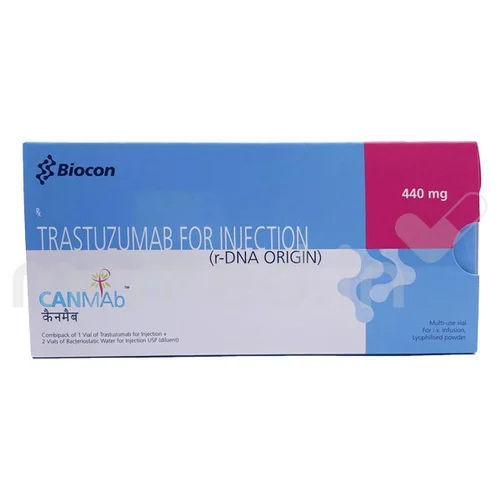
Price 13000 INR/ Unit
- Minimum Order Quantity
- 100 Units
- Supply Ability
- 5000 Units Per Month
- Delivery Time
- 7-10 Seconds
- Main Domestic Market
- All India
- Main Export Market(s)
- Asia, Australia, North America, Eastern Europe, Western Europe

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Anti Cancer Injection Category
Borviz 2.5 Mg Injection
Price 1000 INR / Unit
Minimum Order Quantity : 1 Unit
Shelf Life : upto 24 Days
Fermentation Smell : Normal Smell
Storage Instructions : Dry place
Cytoblastin Aqueous Injection
Price 1000 INR / Unit
Minimum Order Quantity : 10 Units
Shelf Life : upto 24 Months
Fermentation Smell : Normal Smell
Storage Instructions : Dry place
Ph Level : Approximately neutral (pH 3.55 range)
Aravon Edaravone 1.5mg-Ml Injection
Price 1000 INR / Unit
Minimum Order Quantity : 1 Unit
Shelf Life : upto 24 Months
Fermentation Smell : Normal Smell
Storage Instructions : Dry place
Ph Level : Approximately 7.08.0
Doxorubicin Hydrochloride Injection
Price 1000 INR / Unit
Minimum Order Quantity : 1 Unit
Shelf Life : upto 24 Days
Fermentation Smell : Normal Smell
Storage Instructions : Dry place
Ph Level : 2.5 3.5